Today’s cataract surgery is extremely successful at improving patients’ vision and quality of life. Yet, a small percentage of patients remain dissatisfied after the procedure, even if it results in 20/20 visual acuity. One study found the primary cause of post-surgical dissatisfaction in a normal pseudophakic population was dysphotopsias—virtually unknown two decades ago.1
Dysphotopsias are still only vaguely familiar to most optometrists; however, with their incidence on the rise, it is important for us to be able to comanage dysphotopsia cases effectively with surgeons when intervention is necessary.
The term dysphotopsia is used to describe a variety of visual symptoms that result from light reflecting off the intraocular lens (IOL) onto the retina.2 Dysphotopsias are generally divided into two categories: positive and negative. Positive visual changes involve symptoms of bright artifacts, while negative dysphotopsias are perceived as shadows or dark areas in the visual field.3 Patients may report glare, starbursts, halos or shadows when describing their visual symptoms.
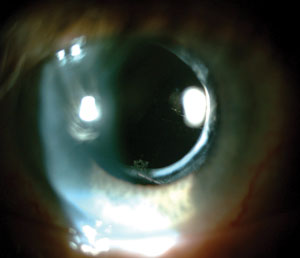 | |
| Introduction of the acrylic intraocular lens material helped to significantly reduce the rate of posterior capsular opacification after cataract surgery. |
It’s difficult to estimate the prevalence of dysphotopsias. Studies report a range from as low as 1.5% to as high as 67% for positive dysphotopsias, with most data showing more moderate numbers of 12% to 35%).4-6 Negative dysphotopsias are less prevalent and are thought to occur in only 0.5% to 2.4% of patients.5,7 In the vast majority of cases, the symptoms subside several weeks after surgery due to the process of neuroadaptation. Neuroadaptation can occur in response to an unwanted monocular or binocular visual disturbance.8 Inherent neural plasticity of the brain helps the visual cortex negate the effect of an undesirable pattern.8,9 This may explain why long-term significant effects are only observed in a small percentage of the pseudophakic population.
IOL Evolution
To understand the increase in the incidence of positive dysphotopsias, it is important to consider the evolution of IOLs. The first commonly accepted intraocular lens material was polymethyl methacrylate (PMMA), which usually coincided with a rounded-edge lens design. Advantages of PMMA material included low cost and durability inside the eye.10 PMMA lenses cause little or no dysphotopsias, which is supported by the fact that dysphotopsias were virtually unknown when PMMA was the IOL material of choice.2 These lenses are rarely used today due to the material’s inability to fold, requiring a large incision during surgery, and a high rate of posterior capsular opacification (PCO) due to edge design.11
Because larger incisions increase the risk of infection and iris complications, and researchers found that the rounded-edge design is a prominent risk factor for PCO, flexible acrylic and silicone materials with vertical, sharp-edged designs were introduced, with great success. Because rising evidence indicates that IOLs with sharp edges result in lower rates of PCO than those with round edges, sharp-edged lens designs are now used more frequently than round-edged designs.12
Acrylic lens material redefined cataract surgery with its ability to fold and fit through small incisions, while also greatly reducing the rate of PCO.13,14 Because of these advantages, acrylic IOLs are the most commonly implanted lenses in the United States today. However, acrylic lens materials has created a subsequent trend of increased incidence of dysphotopsias.2
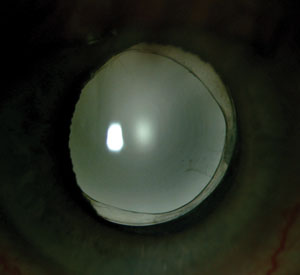 | |
| It’s still unclear if anterior capsular fibrosis increases or reduces negative dysphotopsias. Some clinicians see it as the cause, and use YAG capsulotomy to relieve patients’ symptoms. Others believe that increased opacification and translucency of the lens capsule helps to scatter light into the region of the retina in which the shadow is formed. |
Positive Dysphotopsias
Several studies evaluated the role of IOL material in the formation of positive dysphotopsias. Investigators speculate that positive dysphotopsias are caused by stray light projecting onto the retina, which worsens if that stray light is concentrated in one particular area. One study looked at the light reflected from the surfaces of IOLs and found that when the initial light hits the retina and scatters out of the eye, some of it gets reflected from the posterior surface of the IOL back onto the retina.15 The researchers found that materials with higher refractive indices help to concentrate a larger amount of light onto a smaller area of retina, resulting in symptoms. Acrylic lenses typically have a higher refractive index than PMMA or silicone lenses, further supporting the researchers’ findings.
Another study argues that the increased surface reflectivity of acrylic lenses causes more symptoms compared with silicone or PMMA lenses.16 The reserchers supported their claim by replacing acrylic IOLs of eight symptomatic patients with silicone or PMMA IOLs, which alleviated symptoms of dysphotopsia. Other case reports have similar findings, but no large-scale studies have been done.4,16
In addition to lens material, IOL edge design has also been implicated in the development of positive dysphotopsias.17 Dysphotopsias were a rare occurrence when round-edged designs were used, but an industry move toward sharp edges resulted in higher incidences of dysphotopsias. Using computer analysis, researchers found that both sharp- and round-edged IOLs produce stray light, but only the sharp-edged design concentrated the rays of stray light into an arc on the retina, causing symptoms.18 Round-edged designs spread the light more uniformly throughout the retina, minimizing the effect.
Negative Dysphotopsias
Negative dysphotopsia is a much less studied and understood visual complication than positive dysphotopsia. Patients usually complain of a dark shadow in the temporal visual field. It typically manifests after in-the-bag posterior chamber IOL implantation.19 Numerous theories attempt to identify a cause for negative dysphotopsia; suspects include IOL parameters and optics, corneal incision scars, anterior capsulotomy edge involvement, and distance of IOL from the iris.7,16,17,20
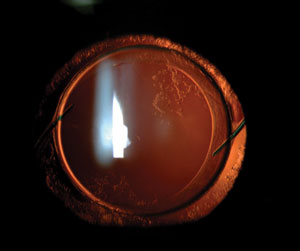 | |
| In-the-bag IOL with mild PCO. Some surgeons improved certain patients’ negative dysphotopsias by replacing an in-the-bag IOL with a sulcus-fixated IOL. Negative dysphotopsia symptoms were never eliminated simply by placing a different IOL inside the capsular bag in place of the original. |
One of the most promising studies used computer software to simulate negative dysphotopsias in normal eye models.19 Researchers found that shadows formed in the peripheral retina when light rays underwent changes at the posterior sharp edge of the lens. Computer analysis showed the formation of ring-patterned scotomas, which would be apparent only temporally on a patient’s visual field, as the nose blocks the nasal aspect.
The researchers determined that several optical factors are required for the formation of negative dysphotopsias.19 The most significant of those factors are: a small pupil; a placement of an acrylic lens at a distance of more than 0.06mm but less than 1.23mm behind the pupil; a sharp-edged design; and a functional nasal retina that extends anterior to the shadow. Secondary factors include a high index of refraction of IOL material and the nasal location of the pupil relative to the eye’s optical axis.19
Several different treatments to reduce negative dysphotopsia have been attempted. Investigators reported that an IOL exchange with a reduction of iris-IOL distance helped reduce symptoms of negative dysphotopsia.20 Similar findings were observed in another study, which found that exchanging an in-the-bag IOL for a sulcus-fixated lens resulted in resolution of symptoms in five eyes of five different women.21 Research also shows that reverse optic capture and secondary piggyback IOL implantation can resolve negative dysphotopsia symptoms.22
Several studies report partial or complete resolution of symptoms after YAG laser anterior capsulotomy of the nasal portion of the anterior capsule.23,24 This evidence disagrees with an earlier study’s hypothesis that eventual opacification and translucency of the anterior capsule helps reduce shadow perception by scattering light into that region of the retina.19 Compared to other studies, this explanation addresses why the incidence of negative dysphotopsias decreases drastically two to three years after surgery. Negative dysphotopsias are still poorly understood, and more studies are needed on this topic.
Management
Dysphotopsias are the primary source of patient dissatisfaction after cataract surgery.1 In most cases they diminish with time, but some patients have severe long-term symptoms.7 It is important for optometrists to recognize dysphotopsias, as we are integral to patient education and comanagement with a surgeon. Case history should be conducted very carefully, beginning with open-ended questions. Probing for specific symptoms may elicit a positive response by certain patients, even in the absence of significant problems. Hyper-focusing on those problems may make symptoms subjectively more bothersome to patients.
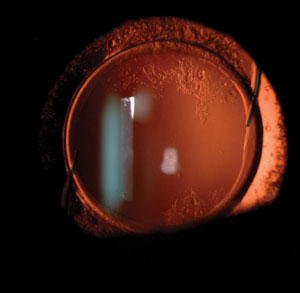 | |
| Intraocular lens exchange may be difficult months or years after the original cataract surgery, as haptics of certain lenses may erode through the edge of the capsule and their manipulation may damage the zonules. |
It is vital to understand that cataract surgery should not be regarded as a “cookie-cutter” treatment that can solve all visual problems of all patients. Individual eyes may respond to surgery in a unique way—some of them by developing visually significant dysphotopsias.
There is currently no agreed-upon management strategy for positive dysphotopsia symptoms. Eye care providers should educate patients that in most cases, they subside after the initial postoperative period, and visual disturbances should disappear without further treatment.7
If severe symptoms persist after four to six weeks, intraocular lens exchange can be considered; however, it should be the last resort. Timing is important when considering an IOL exchange, and earlier intervention may be easier for the surgeon and involves less risk for the patient. Haptics of certain lenses may become fibrosed or start eroding through the edge of the capsule into the sulcus, even several weeks post-surgery.25 Intraocular lenses may also develop a strong adherence to the capsule, making it difficult for the surgeon to dissect it from the capsular bag.26 If IOL exchange is done soon enough after the initial cataract extraction, the surgeon may use the original clear corneal incision.25 Note that if an intraocular lens exchange is considered, YAG capsulotomy should be avoided, as the open posterior capsule requires a vitrectomy during the IOL exchange procedure.25
Some patients with severe negative dysphotopsias have found relief from IOL exchange with reverse optic capture, sulcus fixation and piggyback IOL insertion.20-22 YAG anterior capsulotomy has also been shown to help resolve symptoms of negative dysphotopsias.23,24
Much is still to be studied in this area of cataract surgery, but awareness of these complications is important, as they can cause significant patient dissatisfaction.
Dr. Sukhovolskiy completed his residency at the Jonathan M. Wainwright VAMC and Pacific Cataract and Laser Institute in Kennewick, Wash.
1. Kinard K, Jarstad A, Olson R. Correlation of visual quality with satisfaction and function in a normal cohort of pseudophakic patients. J Cataract Refract Surg. 2013;39:590-7.2. Schwiegerling J. Recent developments in pseudophakic dysphotopsia. Curr Opin Ophthalomol. 2006;17:27-30.
3. Hood CT, Sugar A. Subjective complaints after cataract surgery: common causes and management strategies. Curr Opin Ophthalmol. 2015;26:45-9.
4. Ellis MF. Sharp-edged intraocular lens design as a cause of permanent glare. J Cataract Refract Surg. 2001;27:1061-4.
5. Meacock WR, Spalton DJ, Khan S. The effect of texturing the intraocular lens edge on postoperative glare symptoms: a randomized prospective, double-masked study. Arch Ophthalol. 2002;120:1294-8.
6. Tester R, Pace NL, Samore M, Olson RJ. Dysphotopsia in phakic and pseudophakic patients: incidence and relation to intraocular lens type. J Cataract Refract Surg. 2000;26:810-6.
7. Osher RH. Negative dysphotopsia: long-term study and possible explanation for transient symptoms. J Cataract Refract Surg. 2008;34:1699-1707.
8. Kershner RM. Neuroadaptation and premium IOLs: What does the brain think? Ophthalmology Management. 2011;15(12):51-4.
9. Kent C, Kershner RM, Mainster M, McDonald JE. Multifocal neuroadaptation: Can training help the brain? Rev Ophthalmol. 2010;17(3):24-31.
10. Bellucci R. An introduction to intraocular lenses: material, optics, haptics, design and aberration. Cataract. ESASO Course Series. Basel, Karger, 2013;3:38-55.
11. Auffarth GU, Brezin A, Caporossi A, et al. Comparison of Nd:YAG capsulotomy rates following phacoemulsification of PMMA, silicone, or acrylic intra-ocular lenses in four European countries. Ophthalmic Epidemiol. 2004;11(4):319-29.
12. Cheng J, Wei R, Cai J, et al. Efficacy of different intraocular lens materials and optic edge designs in preventing posterior capsular opacification: a meta-analysis. Am J Opthalmol. 2007;143:428-36.
13. Hollick EJ, Spalton DJ, Ursell PG, et al. The effect of polymethylmethacrylate, silicone, and polyacrylic intraocular lenses on posterior capsular opacification 3 years after cataract surgery. Ophthalmology. 1999;106:49-54.
14. Yan Q, Perdue N, Sage EH. Differential responses of human lens epithelial cells to intraocular lenses in vitro: hydrophobic acrylic versus PMMA or silicone discs. Graefe’s Arch Clin Exp Ophthalmol. 2005;243:1253-62.
15. Erie JC, Bandhauer MH, McLaren JW. Analysis of postoperative glare and intraocular lens design. J Cataract Refract Surg. 2001;27:614-21.
16. Davison JA. Positive and negative dysphotopsia in patients with acrylic intraocular lenses. J Cataract Refract Surg. 2000;26:1346-55.
17. Peng Q, Visessook N, Apple DJ, et al. Surgical prevention of posterior capsule opacification. Part 3: Intraocular lens optic barrier effect as a second line of defense. J Cataract Refract Surg. 2000;26:198-213.
18. Holladay JT, Lang A, Portney V. Analysis of edge glare phenomena in intraocular lens edge designs. J Cataract Refract Surg. 1999;25:748-52.
19. Holladay JT, Zhao H, Reisin CR. Negative dysphotopsia: The enigmatic penumbra. J Cataract Refract Surg. 2012;38:1251-65.
20. Vamosi P, Csakany B, Nemeth J. Intraocular lens exchange in patients with negative dysphotopsia symptoms. J Cataract Refract Surg. 2010;36:418-24.
21. Burke TR, Benjamin L. Sulcus-fixated intraocular lense implantation for the management of negative dysphotopsia. J Cataract Refract Surg. 2014;40:1469-72.
22. Masket S, Fram NR. Pseudophakic negative dysphotopsia: Surgical management and new theory of etiology. J Cataract Refract Surg. 2011;37:1199-1207.
23. Cooke DL, Kasko S, Platt LO. Resolution of negative dysphotopsia after laser anterior capsulotomy. J Cataract Refract Surg. 2013;39:1107-9.
24. Folden DV. Neodymium:YAG laser anterior capsulectomy: Surgical option in the management of negative dysphotopsia. J Cataract Refract Surg. 2013;39:1110-5.
25. Grayson, D. The ins and outs of lens explantation. Rev Ophthalmol. 2011;18(10):36.
26. Leysen I, Bartholomeeusen E, Coeckelbergh T, Tassignon MJ. Surgical outcomes of intraocular lens exchange. Five-year study. J Cataract Refract Surg. 2009;35:1013-8.

