A 47-year-old white woman presented complaining of sudden blurred vision in her left eye. It had persisted for three days and was steadily getting worse. She was using unknown prophylactic medications for migraine, but was otherwise healthy.
A 47-year-old white woman presented complaining of sudden blurred vision in her left eye. It had persisted for three days and was steadily getting worse. She was using unknown prophylactic medications for migraine, but was otherwise healthy.
Her best-corrected visual acuity was 20/15 O.D. and 20/80 O.S. Her pupils were normal, with no afferent defect. There were no external abnormalities on biomicroscopic exam.
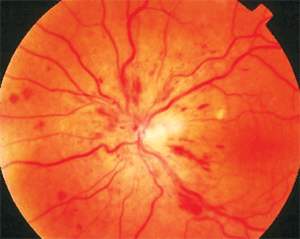
The dilated fundus examination, however, revealed the cause of her symptoms. Her right eye was normal, but her left eye had retinal hemorrhages throughout the four quadrants, as well as disc edema and dilated, tortuous retinal veins.
She was diagnosed with a non-ischemic central retinal vein occlusion (CRVO). This month’s column examines the most recent research about the diagnosis and management of CRVO.
What We Know
CRVO is often an indicator of underlying systemic disease.
The pathogenesis of CRVO is thrombotic obstruction of the central retinal vein as it constricts through the lamina cribrosa. This may involve abnormal blood flow or blood constituents, atherosclerosis and/or vessel anomalies. Properties of blood and the vein itself act in concert to cause thrombus formation, which impedes venous blood flow from the retina.1-4
CRVO often is an indicator of underlying systemic disease, such as diabetes, hypertension, atherosclerosis or various coagulopathies.2,4 Refer patients with CRVO and unknown systemic health to an internist to look for vasculopathic risk factors.
A New Association
While vasculopathic diseases are common associations and precipitating conditions for CRVO in elderly patients, they are not often found in association with CRVO in younger patients, specifically under the age of 50 years. Rather, in these patients, coagulopathies and thrombophilic disorders are more common.
A significant consideration and associated finding in younger patients with CRVO is antiphospholipid antibody syndrome (APAS).5 Up to 10% of healthy individuals have circulating antiphospholipid antibodies.5 Approximately 50% of patients with antiphospholipid antibodies also have systemic lupus erythematosus (SLE) or other autoimmune diseases.5 In many cases, however, APAS exists without associated autoimmune disease.5,6
Antiphospholipid antibodies are a group of circulating antibodies that includes anticardiolipin antibody, lupus anticoagulant and the Biologic False Positive Test for Syphilis (BFP-TS). These antibodies are directed against phosopholipid-binding proteins, which prolong phospholipid-dependent coagulation assays.5
In APAS, the body erroneously identifies cell membrane phospholipids as foreign, and consequently, it produces antibodies. These antibodies appear to have an affinity for the cell membranes found in platelets, vessel endothelial cells and clotting factors.5-7
Antiphospholipid antibodies (anticardiolipin antibodies and lupus anticoagulant) commonly occur in patients with SLE.6,7 When these antibodies occur in the presence of SLE, it is considered a secondary syndrome. In 1985, Graham R.V. Hughes, M.D., F.R.C.P., identified these antibodies in seemingly normal patients who experienced thrombotic events and pregnancy loss in the absence of SLE.8
Antiphospholipid antibody syndrome is diagnosed by arterial and venous thrombosis, pregnancy loss and/or thrombocytopenia in the presence of lupus anticoagulant and anticardiolipin antibodies.9 The primary form exists in the absence of clinically or serologically proven autoimmune disease and has been termed primary antiphospholipid antibody syndrome (PAPAS), or Hughes’ syndrome.
The most common conditions that result from primary APAS and affect the eye and visual system are arterial or venous thrombosis with resultant ischemia. This manifests as CRVO or central retinal artery occlusion (CRAO), anterior ischemic optic neuropathy (AION), migraine, ophthalmoparesis and diplopia, amaurosis fugax, retinal hemorrhages and cotton-wool spots, and retinal neovascularization.10-12
While rheumatologists and other primary care physicians are mindful that patients with PAPAS can have ocular manifestations, many ophthalmic practitioners often don’t associate ocular thrombosis with PAPAS, and thus, don’t test for these antibodies or suggest its inclusion in a diagnostic evaluation.
Antiphospholipid antibody syndrome must be considered in cases of retinal vascular occlusion and other thrombotic conditions in younger patients (under age 50). It should especially be considered in unusual thrombotic events, such as recurrent or bilateral artery or vein occlusion, or combined artery and vein occlusion (especially if there is no history of vascular disease), particularly if the patient is young.
Treatment Studies for CRVO
• SCORE. The Standard Care vs. Corticosteroid for Retinal Vein Occlusion (SCORE) CRVO study evaluated the effectiveness of intravitreal triamcinolone (either 1mg or 4mg) in patients with macular edema secondary to a perfused central retinal vein occlusion.13 The study also compared steroid treatments to the current standard of care for CRVO: observation without intervention.
After 12 months, 27% of patients who received a 1mg steroid injection and 26% who received a 4mg injection achieved the desired visual improvement—a gain in visual acuity of 15 or more letters. By comparison, just 7% of the patients who were observed without treatment achieved the desired visual outcome. The study investigators concluded that intravitreal triamcinolone was superior to observation for treating vision loss associated with macular edema secondary to CRVO.
Both intravitreal steroid groups showed similar effectiveness, but the 1mg participants experienced fewer adverse effects. At this point, it seems that a 1mg dose of intravitreal triamcinolone should be considered anywhere from onset to one to two years to improve vision loss secondary to macular edema following CRVO.13
• CRUISE. A 12-month prospective multi-center study, CRUISE is intended to assess the safety, tolerability and efficacy of intravitreal Lucentis (ranibizumab, Genentech) injections vs. placebo in the treatment of macular edema secondary to CRVO. Study endpoints include visual acuity improvement, as well as anatomic reduction in macular thickness and edema. Patients received a placebo injection, or 0.3mg or 0.5mg of ranibizumab on a monthly basis for six months. During the second six-month period, patients were evaluated monthly and reinjected as needed. The trial has not yet been completed, so there are no published results yet. However, preliminary data presented at Retina Congress 2009 in New York indicated success at the six-month mark. Patients who received 0.3mg of Lucentis had a mean visual improvement of 12.7 letters, and patients who received 0.5mg of Lucentis achieved a mean improvement of 14.9 letters. Decreased macular thickness on optical coherence tomography was greater in both treatment arms than in patients receiving placebo.
Response time also seems very significant. Patients in the treatment arms demonstrated a decrease in central foveal thickness, with a reduction of 400µm on day seven. By comparison, the placebo injection group showed a reduction in central foveal thickness of just 167.7µm.
We referred the patient for medical evaluation with special emphasis on hypertension, diabetes, hyperviscosity and antiphospholipid antibody elevation. It turned out that she did have elevated antiphospholipids, signaling antiphospholipid antibody syndrome. She is undergoing further evaluation with consideration for ongoing antithrombotic therapy.
1. Wang GL, Zhang F, Peng XY, et al. The study of clinical characteristic of nonischemic central retinal vein occlusion. Zhonghua Yan Ke Za Zhi. 2008 Feb;44(2):152-6.
2. O’Mahoney PR, Wong DT, Ray JG. Retinal vein occlusion and traditional risk factors for atherosclerosis. Arch Ophthalmol. 2008 May;126(5):692-9.
3. Klatt C, Purtskhvanidze K, Hasselbach H, et al. Retrospective case analysis of ophthalmological and systemic risk factors in patients with retinal vascular occlusion. Ophthalmologe. 2009 Sep 16. [Epub ahead of print].
4. Di Capua M, Coppola A, Albisinni R, et al. Cardiovascular risk factors and outcome in patients with retinal vein occlusion. J Thromb Thrombolysis. 2009 Aug 25. [Epub ahead of print].
5. Lim W, Crowther MA, Eikelboom JW. Management of antiphospholipid antibody syndrome: a systematic review. JAMA. 2006 Mar 1;295(9):1050-7.
6. Marai I, Levi Y, Godard G, et al. Following 90 patients with antiphospholipid syndrome with antibody titers and correlations with clinical manifestations: symptoms of the disease, a new antibody and correlations with clinical manifestations in the Israeli population. Harefuah. 2001 Jun;140(6):495-500, 565.
7. Mackworth-Young CG, Loizou S, Walport MJ. Primary antiphospholipid syndrome: features of patients with raised anticardiolipin antibodies and no other disorder. Ann Rheum Dis. 1989 May;48(5):362-7.
8. Hughes GR. The anticardiolipin syndrome. Clin Exp Rheumatol 1985 Oct-Dec;3(4):285-6.
9. Cuadrado MJ, Khamashta MA. The anti-phospholipid antibody syndrome (Hughes syndrome): therapeutic aspects. Baillieres Best Pract Res Clin Rheumatol. 2000 Mar;14(1):151-63.
10. Au A, O’Day J. Review of severe vaso-occlusive retinopathy in systemic lupus erythematosus and the antiphospholipid syndrome: associations, visual outcomes, complications and treatment. Clin Experiment Ophthalmol. 2004 Feb;32(1):87-100.
11. Suvajac G, Stojanovich L, Milenkovich S. Ocular manifestations in antiphospholipid syndrome. Autoimmun Rev. 2007 Jun;6(6):409-14.
12. Dori D, Beiran I, Gelfand Y, et al. Multiple retinal arteriolar occlusions associated with coexisting primary antiphospholipid syndrome and factor V Leiden mutation. Am J Ophthalmol. 2000 Jan;129(1):106-8.
13. Ip MS, Scott IU, VanVeldhuisen PC, et al. SCORE Study Research Group. A randomized trial comparing the efficacy and safety of intravitreal triamcinolone with observation to treat vision loss associated with macular edema secondary to central retinal vein occlusion: the Standard Care vs Corticosteroid for Retinal Vein Occlusion (SCORE) study report 5. Arch Ophthalmol. 2009 Sep;127(9):1101-14.