A
weekly e-journal by Art Epstein, OD, FAAO
Off the Cuff: Best of Clinical Excellence Awards 2019 – Part 1
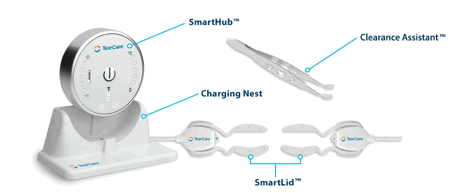
I frequently am asked what I use, how I use it and why. So, every now and then, especially when I discover new devices, drugs or instruments, I put together a "What’s what" of notable advances. It's been quite a while since my last one. Since tear dysfunction and ocular surface disease are my primary focus, Part 1 will cover that area with more to come. Note that some of what I am covering may not be new, but it’s gotten past my clinically conservative NY state of cynicism and I’ve incorporated it within our practice with good success. Here are my picks for new and disruptive technology in no particular order. For the distrusting, I am not being paid to write any of this. I am really excited about Sight Sciences’ TearCare System. It is a clinically excellent and cost-effective treatment system for MGD in an easy-to-implement small package. Unlike LipiFlow, which has long been the gold standard for MGD management, TearCare applies controlled heat externally, allowing the patient to keep their eyes open, and can be easily applied regardless of aperture size. We use both LipiFlow and TearCare, depending on the patient. Tear Care acceptance and clinical performance has been excellent. More on how well this system works to come. For now, highly recommended!
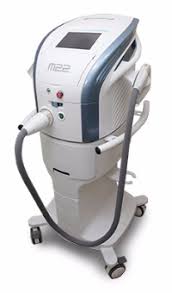
HyClear is a second-generation hypochlorous acid topical spray introduced by Contamac earlier this year. Many of you know I am a huge proponent of HOCl, which directly controls overpopulation of lid flora commonly seen in MGD, OSD and blepharitis. What is all too often missed is that bacterial elaboration of toxins and digestive enzymes, like lipase, are key factors in disrupting tear stability and driving tear dysfunction. What I love about Hyclear is that it is sold only to ECPs—I am a big believer in controlling the patient experience by directly providing what they need. It is also fairly priced for patients and remains stable for well over a year after opening. I have patients spray it directly onto their closed eyelids, flutter for a few seconds to spread it, and wipe it off after 10 seconds. Clinical improvement in signs and symptoms are rapid and impressive. It took quite some time before I took the plunge and decided to incorporate IPL into the practice, but the sheer brilliance of my friend and IPL guru Dr. Laura Periman and a massive amount of research, finally convinced me that IPL is an essential part of a comprehensive dry eye management. We purchased a Lumenis M22 Optima IPL late last year and it has literally transformed the practice. Incredible engineering, overwhelming patent acceptance and clinical performance that has exceeded my most optimistic expectations, I love my M22! Expect to hear more about IPL, photo-modulation and meibomian gland remodeling in the coming months. If you are performing IPL regularly, I highly recommend Opt Slims protective eyewear from Innovative Optics. Opt Slims are a lightweight, sporty frame with digital IPL lenses that automatically darken instantly with each IPL flash to protect eyes and prevent discomfort associated with continued IPL exposure. They are not inexpensive, but they eliminate post pulse afterimages and help make treatment more comfortable and precise. For me, they were well worth it, and Innovative Optics is a pleasure to deal with. Part 2 to come with some surprises...
|
|||||
 |
||
| Correlation Between Type 2 Diabetes, Dry Eye and Meibomian Glands Dysfunction | ||||
Prospective study of 76 males, 37 with type 2 diabetes with an average of duration between 7±5 years, and 36 males from control group to assess the meibomian glands, ocular surface and tear function in patients with type 2 diabetes, and study the correlation between these conditions. After completing an ocular surface disease index (OSDI) questionnaire, the non-invasive tear film break-up time (NIBUT) and the tear lipid layer pattern was performed using interferometry system and tear meniscus height/TMH. Ocular surface was studied with lissamine green staining and morphology of the glands with evaluation of Marx's line (lid margin abnormalities); meibomian secretion was expressed and was also assessed the quality. The results were analyzed using the statistical Kruskal-Wallis and Mann-Whitney, with correlations by Spearman Rho. The mean age was 59±8 years; 71% of participants presented MGD (76% diabetics and 67% controls). OSDI were significantly higher in the diabetic group. A positive correlation was found between glycemia and symptoms, and a strong correlation between Hb1Ac and OSDI in MGD. NIBUT was lower in the control group (2.47±1.2s) than for the diabetic group (2.9±1.2s), with a significant inverse correlation (52.22%) with MG inflammation. MGD in type 2 diabetic patients was more severe compared with nondiabetic patients. Longer duration of diabetes was associated with major symptoms and changes in MG. The diabetic group showed major changes in lids and tear function, accounting for evaporative dry eye and presenting a high degree of correlation with MG inflammation and obstruction. |
||||
SOURCE: Sandra Johanna GP, Antonio LA, Andrés GS. Correlation between type 2 diabetes, dry eye and meibomian glands dysfunction. J Optom. 2019; May 23. [Epub ahead of print]. |
||||
 |
||
| Short-Term Results of Trial of Topical Insulin for Treatment of Dry Eyes in Diabetics | ||||
In this randomized, double-blind interventional study, diabetics with dry eye disease (DED) ages 18 to 60 years were randomly assigned to TI or standard artificial tears (SAT) to assess the short-term effects of topical insulin (TI)—1 unit/drop four times per day for four weeks—on the symptoms and signs of diabetic with DED. Baseline Ocular Surface Disease Index (OSDI), Schirmer I test (ST), tear break-up time (TBUT) and ocular Sjögren's International Collaborative Clinical Alliance (SICCA) score were compared four weeks after treatment. A total of 160 participants (involving 320 eyes) received either TI (n=80) or SAT (n=80). After four weeks of treatment, a significant number of participants in both TI and SAT groups showed improvement in their OSDI score, 66% and 63%, respectively, but were not significantly better than each other. However, most participants in both groups showed worsening of ST and TBUT. Most of the participants in both groups showed no change in their ocular SICCA score (p>0.05). The study showed a significant and similar improvement in the OSDI score for TI 1 unit/drop four times daily and SAT in treating diabetics with DED. Researchers wrote that further research would be required to understand the long-term effects of TI on the ocular surface. |
||||
SOURCE: Aniah Azmi N, Bastion MC. Short-term results of trial of topical insulin for treatment of dry eyes in diabetics. Eye Contact Lens. 2019; May 22. [Epub ahead of print]. |
||||
| Large Field of View Corneal Epithelium and Bowman's Layer Thickness Maps in Keratoconic and Healthy Eyes | ||||
In 47 patients (one eye) with keratoconus (KC) and 20 healthy subjects (one eye), investigators assessed differences in epithelium thickness (ET) and Bowman's layer thickness (BLT) maps in keratoconic eyes and healthy eyes. Epithelium and Bowman's layer measurements were performed with a custom-designed polarization sensitive (PS)-OCT with a conical scanning optics design. En-face corneal ET and BLT maps with a diameter of 11mm were computed. The mean ET and BLT of 25 sectors, the thinnest (minET, minBLT) and the thickest sector (maxET, maxBLT) were assessed. Ratios between thinnest/thickest sector (R1) as well as between mean ET and BLT of the inferior temporal quadrant/the superior nasal quadrant (R2) were calculated (R1ET, R1BLT; R2ET, R2BLT). Receiver operator characteristic (ROC) curve analysis was used to assess the diagnostic power of statistically different parameters. In healthy eyes, smooth ET maps were observed. KC eyes showed a "doughnut pattern." The BLT maps of healthy eyes had a smooth appearance, but a highly irregular "moth damage"-like pattern could be observed in keratoconic eyes. Highest AUC values were found for the thinnest sector of the BLT map, R1ET and the thinnest sector of the ET-map. PS-OCT imaging enabled the visualization of significant differences of the corneal epithelium and the Bowman's layer in en-face maps covering almost the entire cornea. Investigators wrote that epithelial thickness and BLT profiles in this study showed their diagnostic importance for distinguishing keratoconic and healthy eyes. |
||||
SOURCE: Pircher N, Beer F, Holzer S, et al. Large field of view corneal epithelium and Bowman's layer thickness maps in keratoconic and healthy eyes. Am J Ophthalmol. 2019; Jun 3. [Epub ahead of print]. |
||||
| News & Notes | ||||||||||||||||||||||
| Allegro Phase II Study Evaluating Risuteganib Meets Primary Endpoint Allegro Ophthalmics announced that its Phase II study of risuteganib (Luminate) for the treatment of intermediate nonexudative age-related macular degeneration met its primary endpoint, with 48 percent of patients in the risuteganib arm gaining ≥8 letters of vision at week 28 compared with baseline. The primary endpoint was the proportion of subjects with a ≥8 letters of vision gain with two risuteganib injections vs. one sham treatment. The trial was a prospective, randomized, double-masked, placebo-controlled multicenter study that evaluated the safety and efficacy of risuteganib. Read more. |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
Optometric Physician™ (OP) newsletter is owned and published by Dr. Arthur Epstein. It is distributed by the Review Group, a Division of Jobson Medical Information LLC (JMI), 11 Campus Boulevard, Newtown Square, PA 19073. HOW TO ADVERTISE |